VST
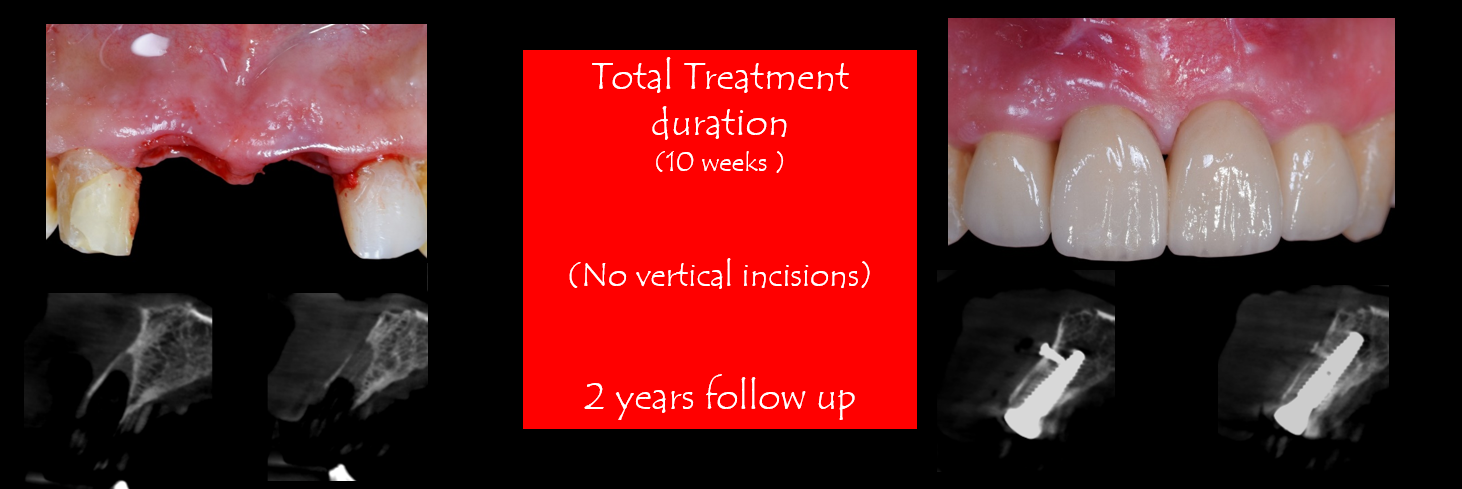
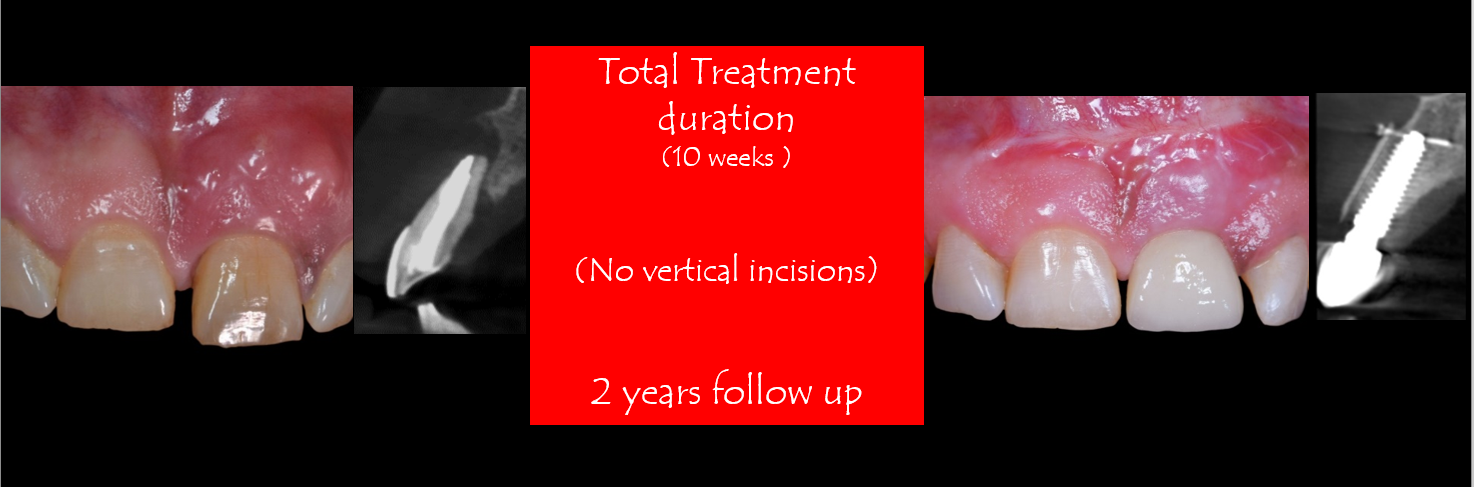
The Vestibular Socket Therapy: A Novel approach for immediate implant placement in defective fresh extraction sockets with or without active socket infection
Elaskary Micro-Surgery Kit
Elaskary Micro-Surgery Kit (Storz am Markt GMBH, Emmingen-Liptingen, Germany) the instruments were designed to achieve maximum comfort for grasping, thus allows better tissue access, and less tissue trauma. The kit consists of Micro bald holder used for precise micro incisions next to the inter-dental papilla and a micro periosteal elevator (Mini-Buser), tissue forceps 0.6mm, straight, 17.5cm straight and curved micro-scissors that are serrated and 16 cm Gomel & curved,as well as Micro needle holder Barraquer TC, 0.8mm, curved, 18cm, with smooth Carbide tips that is recommended for 5-0 to 8-0 suturing.

Elaskary Micro-Surgery Kit
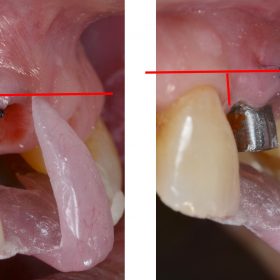
Elaskary Titanium Papillary Insert (TPI)
Elaskary Titanium Papillary Insert (TPI) is composed of a pyramidal-shaped, polished titanium core 2 to 3 mm in height, 1 mm in width, and 3 mm in length. It was designed as a foundational support for the development of interim-plant papillae; it yields immediate results, and it is thought to allow for a predictable and stable esthetic outcome. The TPI is placed between adjacent implants, and it requires more than 3 mm of inter-implant distance to ensure sufficient blood supply. A 1-mm diameter twist drill is used to create a 5-mm deep osteotomy, and then the insert is screwed in with cotton pliers until it is flushed with the alveolar crest. Primary soft tissue closure is obtained and healing ensues.
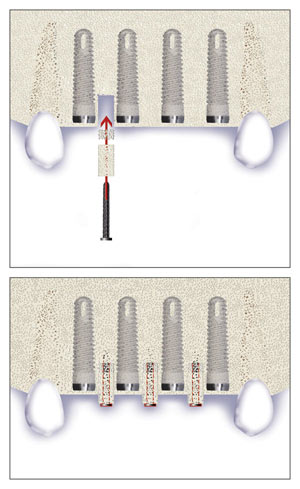
Elaskary Titanium Papillary Insert (TPI)
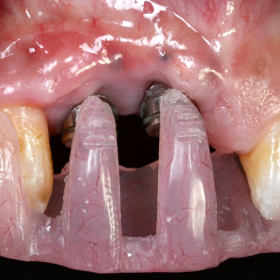
The Inter-Implant Papilla
A technique has been introduced by Dr. Elaskary in 1999. The Inter-Implant Papilla regenerative template1 is a carrier fabricated from pure titanium. It acts as a housing for any bone grafting material that is to be placed on the alveolar ridge, to regenerate an osseous foundation for the inter-implant papilla. The template is to be placed at the time of implant insertion, therefore eliminating the need for any additional surgical procedure. Other advantages of the template are that it carries and protects the bone graft material and also separates the bone grafting mix from the undesired fibroblast and epithelial cells, which favors graft predictability. The use of the template requires a space of not less than 3 mm between two adjacent implants. After the implants are inserted, the inter-implant bone is decorticated, to allow for a pool of cellular activity to the graft. The preferred grafting mix used with the template is 50% autogenous bone chips harvested from the drilling procedure and 50% allograft bone chips (Porous, Zimmer dental, Carlsbad, U.S.A.). However, the use of 100% autogenous chips can provide a more predictable clinical outcome but, is liable for greater post operative remodeling and resorption. The template is then placed on the alveolar ridge with its two perforated ends facing the sides of the alveolar ridge. Two GBR fixation pins (Autotac system, Biohorizons, Alabama, U.S.A.) are fitted into the perforated ends of the template to stabilize the template and secure it in place. Soft tissue will require some manipulations in order to achieve complete tension free closure which is of extreme value to the template survival. At the time of the second-stage surgery, the template is removed, revealing the regenerated bone, and the flap is then sutured and left to heal. After soft tissue healing is complete, the provisional prosthesis might be used to develop a natural emergence profile. Care must be exercised to use a smooth & thin provisional prosthesis margins in order not to exert any pressure on the newly formed bone.

The Inter-Implant Papilla
Recession Template
This template consists of an acrylic extension resting on the palatal surfaces of the maxillary teeth adjacent to the dental implant with a buccal flange projecting onto the labial surface, stopping short of the level of the receded gingival margin. During the review visits, the acrylic template would be placed back and the difference in the labial gingival margin can then be measured through the labial flange.